Drug plans decoded: Biosimilars Part 3

Public plans drive adoption of biosimilars
So far, four provinces in Canada —British Columbia, Alberta, Quebec and New Brunswick—have adopted biosimilar transitioning or switching strategies, in their own way and at their own pace.
B.C. was the first to implement a strategy, starting in late 2019 after a six-month transition period; given the province’s universal PharmaCare program, claims data from TELUS Health illustrates a dramatic shift toward biosimilars for public and private plans alike.
Private plans across Canada have also begun to experience the “halo effect” as physicians gradually change their prescribing habits for all patients who require biologic drugs. Some insurance carriers have also implemented programs that align or coordinate with provincial policies.
Broadly speaking, a switching policy requires that patients currently taking a reference biologic transition to one of its approved biosimilars by a certain date to continue to receive public coverage. Exceptions may apply for medical reasons.
This article summarizes the provinces’ approaches to increasing the uptake of biosimilars. Table 1 compares the scope of the switching programs in the four provinces where these programs are in place.
This is the third in a series of articles: article one described the nature of biologics, why interchangeability is not an option and the cost differences between reference biologics and their biosimilars; article two compared Canada to Europe, where many countries are considered leaders in the utilization of both reference and biosimilar biologics.
It’s important to note that a single reference biologic may have multiple transition periods because the policy is based on health indications. For example, in B.C., patients taking the reference biologic of Remicade for rheumatoid arthritis transitioned before those who were taking Remicade for Crohn’s disease.
Table 1 – A comparison of biosimilar switching policies in Canada
| Province | Reference biologics* | Biosimilar biologics | Health indications |
| British Columbia | 5 | 13 | 12 |
| Alberta | 10 | 23 | 11 |
| Quebec | 10 | 27 | Pending |
| New Brunswick | 7 | 18 | 12 |
*Excluding Copaxone, a non-biologic complex drug.
Source: TELUS Health as of October 1, 2021
British Columbia
B.C. launched its Biosimilars Initiative in May 2019; a few months later, in November 2019, its first six-month transition period, affecting three reference biologics and five indications, ended. The province has expanded the program as more biosimilars become available and its fourth transition period recently ended in October 2021. In total at this time, B.C.’s switching policy encompasses five reference biologic drugs (Enbrel, Humira, Lantus, Remicade and Rituxan), 13 biosimilars and 12 indications (mainly autoimmune diseases and diabetes).
The province has reported that 73% of patients in phase one (involving Enbrel, Lantus and Remicade) and 78% of patients in phase two (involving Remicade for two additional indications) switched to a biosimilar. More than 90% of all PharmaCare patients taking a biologic, including those starting on a biologic, are taking a biosimilar.
The B.C. government anticipates savings of close to $100 million after the first three years, which it has stated will be reinvested into health care. For example, it has expanded coverage for diabetes and arthritis drugs, and launched a program for patients with complex inflammatory bowel disease.
TELUS Health claims data confirms that B.C.’s switching policy has boosted claims for biosimilars on the private side as well. In fact, an analysis of four biologic molecules (etanercept, infliximab, insulin glargine and rituximab) reveals the breakdown between reference biologics and biosimilars has essentially flipped. When the government announced the initiative,
B.C.’s universal PharmaCare program has fuelled those results, as some insurance carriers (such as Equitable Life, Green Shield Canada and Pacific Blue Cross) align or coordinate with the Biosimilars Initiative, meaning their patients cannot turn to their private plan to pick up full coverage for a reference biologic that is no longer covered by PharmaCare. Other insurance carriers are picking up full coverage for the reference biologics, after having negotiated discounted pricing with manufacturers.
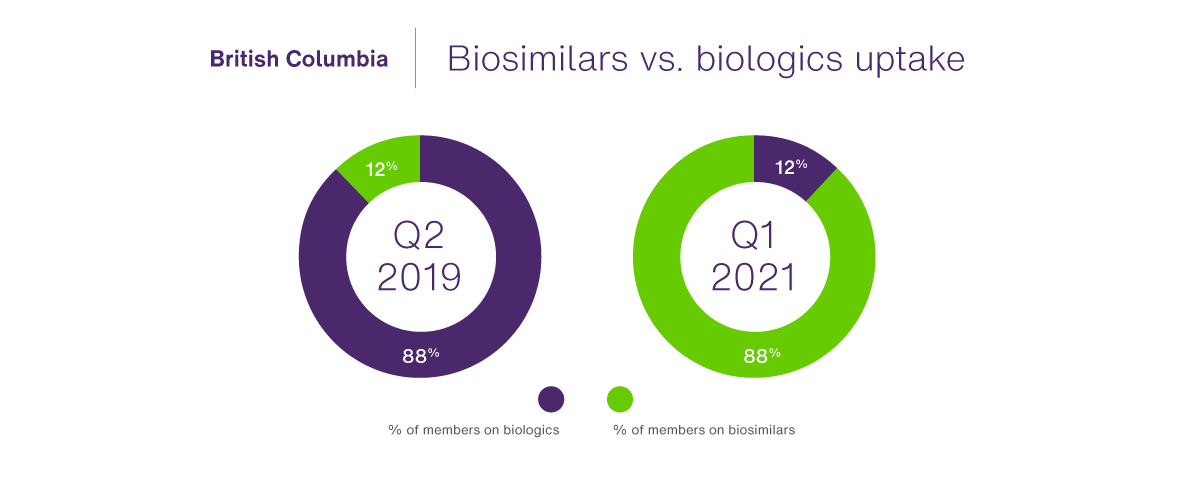
Figure 1 – Breakdown in private coverage between reference and biosimilar biologics in B.C., before and after implementation of the Biosimilars Initiative
Source: TELUS Health claims data for private plans in B.C. for the following biologic drug classes: infliximab, etanercept, insulin glargine, rituximab; Q2 2019 and Q1 2021
Alberta
Alberta announced its Biosimilar Initiative in December 2019 (due to the COVID-19 pandemic, the initial six-month transition period was extended until January 2021). Over the course of 2020, six reference biologics (Enbrel, Lantus, Neulasta, Neupogen, Remicade and Rituxan) for nine indications became part of the program, which also includes the reference drug Copaxone for multiple sclerosis, a non-biologic complex drug. Glatect is the subsequent-entry drug for Copaxone.
So far in 2021, Alberta has added Lovenox, Humira, Humalog and, most recently, NovoRapid to its list of reference biologics affected by its switching policy. Patients have until the following dates to switch: January 10, 2022 for those currently taking Lovenox (for blood clotting disorders); February 1, 2022 for Humalog and April 1, 2022 for NovoRapid (both for diabetes); and May 1, 2022 for Humira (for autoimmune disorders). In total as of November 2, 2021, Alberta’s Biosimilar Initiative encompasses 10 reference biologics, 23 biosimilars and 11 indications.
As in B.C., TELUS Health claims data illustrates a dramatic turnaround in biosimilars uptake for private drug plans. For the three biologic molecules of etanercept, infliximab and insulin glargine, biosimilars’ share of claims jumped from just five percent at the end of the second quarter of 2019 to 72% by the end of the first quarter in 2021 (Figure 2).
The Alberta government anticipates savings of between $227 million and $380 million over four years once the program is fully implemented. It has also stated those savings can be reinvested in health care.
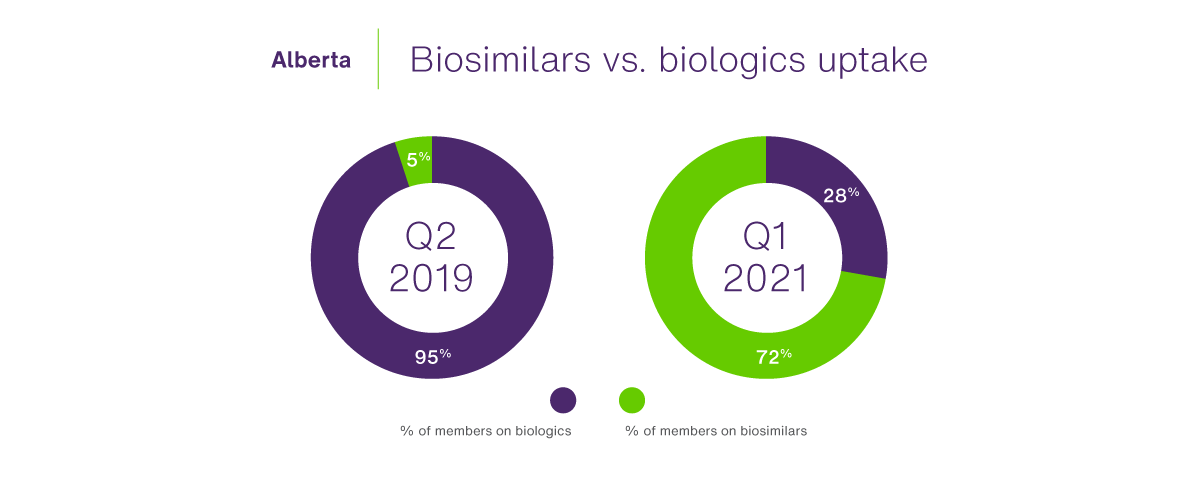
Figure 2 – Breakdown in private coverage between reference and biosimilar biologics in Alberta, before and after implementation of the Biosimilars Initiative
Source: TELUS Health claims data for private plans in Alberta for the following biologic drug classes: infliximab, etanercept, insulin glargine, rituximab; Q2 2019 and Q1 2021
Quebec
On May 18, 2021, Quebec announced that patients taking a reference biologic for which biosimilars are available must switch to a biosimilar by April 12, 2022, to continue to receive coverage from the public drug plan.
Details on its rollout are forthcoming. Based on the province’s current list of biosimilar drugs available for coverage, the program will involve 10 reference biologics and 27 biosimilars. It will also include Copaxone, the reference non-biologic complex drug for multiple sclerosis (as in Alberta).
New Brunswick
New Brunswick launched its Biosimilars Initiative on April 20, 2021. Patients taking the reference biologics of Enbrel, Humalog, Humira, Lantus, Remicade and Rituxan have until November 30, 2021 to switch to a biosimilar. Patients taking Lovenox have until February 28, 2022. In total, seven reference biologics and 18 biosimilars will be part of the program, for treatment of 12 health indications. Additionally, as in Alberta and Quebec, New Brunswick’s switching program includes the non-biologic drug Copaxone.
In a press release, the province stated expected savings of $10 million once implementation is complete, which will be reinvested in coverage of new therapies.
Nova Scotia, Prince Edward Island, Newfoundland and Labrador
The remaining Atlantic provinces have not announced any plans to implement switching policies. Nova Scotia and Prince Edward Island currently provide the same reimbursement criteria and listing status for reference and biosimilar biologics.
In Newfoundland and Labrador, the public plan gives preferential access to biosimilars for new patients.
Saskatchewan
As of November 2, 2021, Saskatchewan has not put a switching policy in place. For new patients, a preferential access approach seeks to encourage the use of biosimilars; however, until recently, new patients could still get coverage for the reference biologic.
That changed on March 1, 2021, when public coverage became limited to biosimilars for patients with a new prescription for etanercept (reference biologic of Enbrel). On June 21, 2021, that policy was extended to include adalimumab (reference biologic of Humira).
Manitoba
Manitoba does not have a switching policy as of November 2021. In August 2018, it implemented a tiered biologics reimbursement policy, which requires new patients to use a biosimilar first. If that treatment fails, patients can try a reference biologic.
Ontario
Ontario has not implemented a switching policy. In early 2020, the government had announced a plan to do so, but the pandemic took priority and no additional announcements have been made since.
Currently, Ontario lists some biosimilars and selected reference biologics in its formulary as limited-use drugs, which makes them easier to prescribe than reference biologics for which coverage is determined on a case-by-case, annual basis through the Exceptional Access Program. For new patients, coverage is limited to biosimilars for approved indications.
Despite the absence of a switching policy, Ontario’s private drug plans appear to be experiencing somewhat of a halo effect in the adoption of biosimilars as prescribing patterns slowly change across the country and as insurance carriers adopt their own national policies. For the biologic molecules of etanercept, infliximab, insulin glargine and rituximab, TELUS Health reports that biosimilars’ share in Ontario grew from just four percent in the second quarter of 2019 to 14% by the end of the first quarter of 2021 (Figure 3). Still, this is well behind the turnarounds in uptake experienced in B.C. and Alberta, where switching policies are well established.
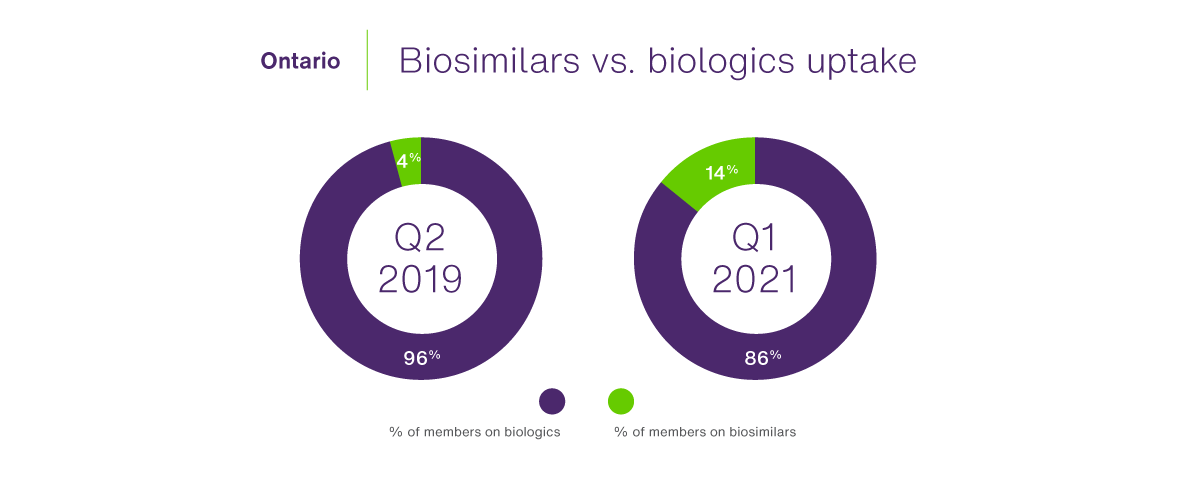
Figure 3 – Breakdown in coverage between reference and biosimilar biologics in Ontario, 2019 versus 2021
Source: TELUS Health claims data for private plans in Ontario for the following biologic drug classes: infliximab, etanercept, insulin glargine, rituximab; Q2 2019 and Q1 2021
In summary
Across Canada, uptake of biosimilars has historically been low despite measures by public plans that encourage existing patients to use them, and require new patients to do so. Biosimilar switching policies dramatically increase adoption, as demonstrated by claims data in two of the four provinces that have implemented such programs since 2019. Private plans in those provinces have also benefited, and to a lesser extent in other provinces, due to the halo effect and changes in policy on the private side.
The landscape for the coverage of biologics in Canada is constantly evolving. Working with their benefits advisor and insurer, plan sponsors of private drug plans can evaluate retrospective and prospective claims data as well as corporate objectives to determine whether to maintain current coverage practices for biologics, align or coordinate with provincial policies, or explore discounted pricing with biologic manufacturers. Our next article will discuss plan sponsors’ options in greater detail.
